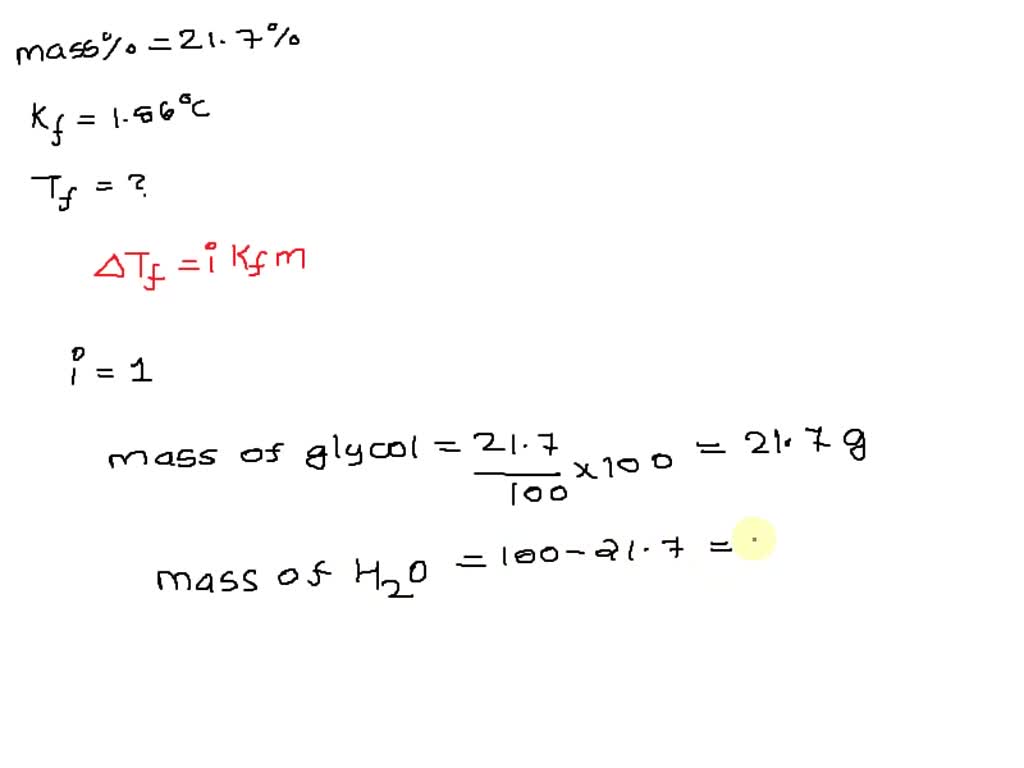Impact of Sc for Fe3O4−C2H6O2−H2O and Fe3O4−Co/C2H6O2−H2O on Hη and Gη... | Download Scientific Diagram

Impact of Q for Fe3O4−C2H6O2−H2O and Fe3O4−Co/C2H6O2−H2O on fη and θη... | Download Scientific Diagram
Calculate the mole fraction of ethylene glycol (C2H6O2) in a solution containing 20% of C2H6O2 by mass. - Sarthaks eConnect | Largest Online Education Community

Impact of Q for Fe3O4−C2H6O2−H2O and Fe3O4−Co/C2H6O2−H2O on fη and θη... | Download Scientific Diagram
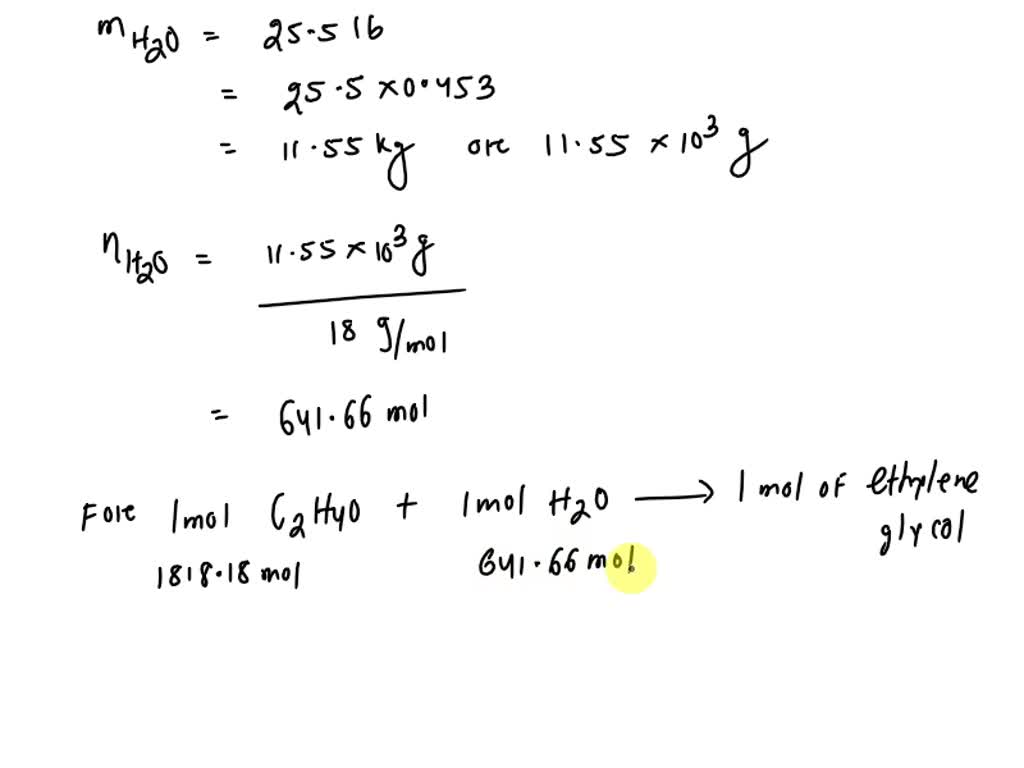
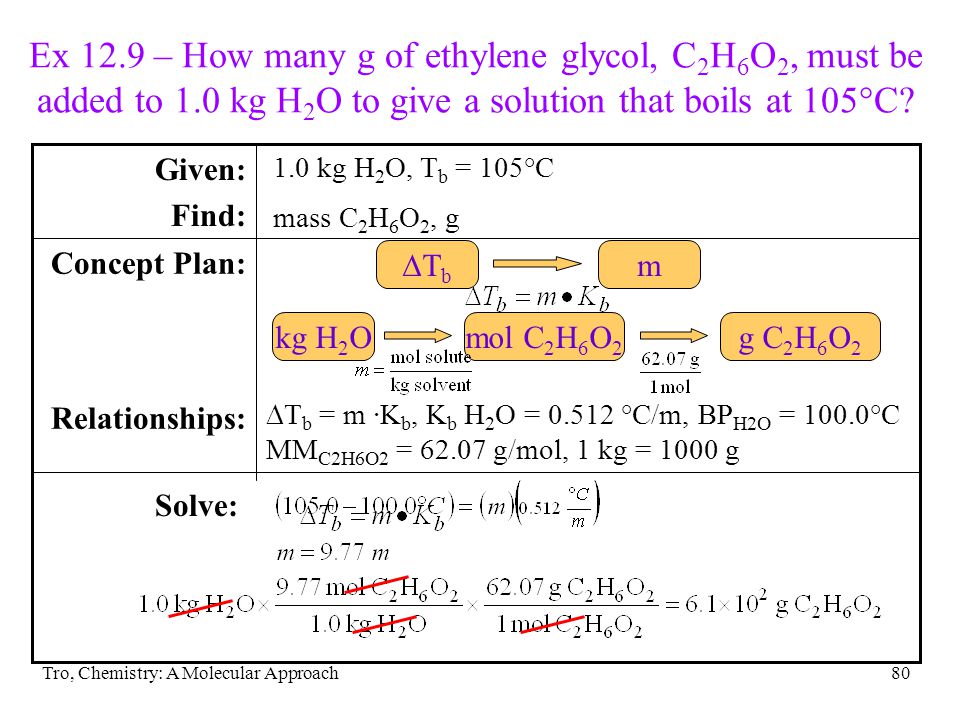
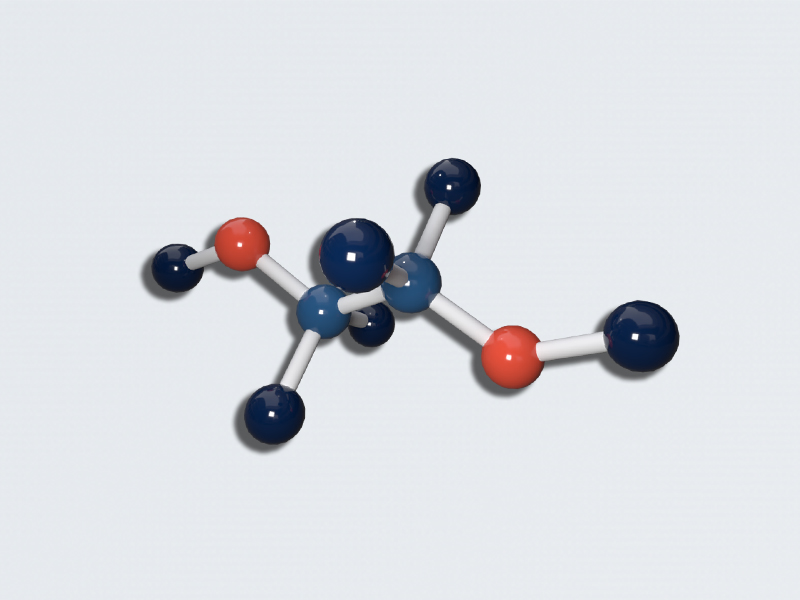
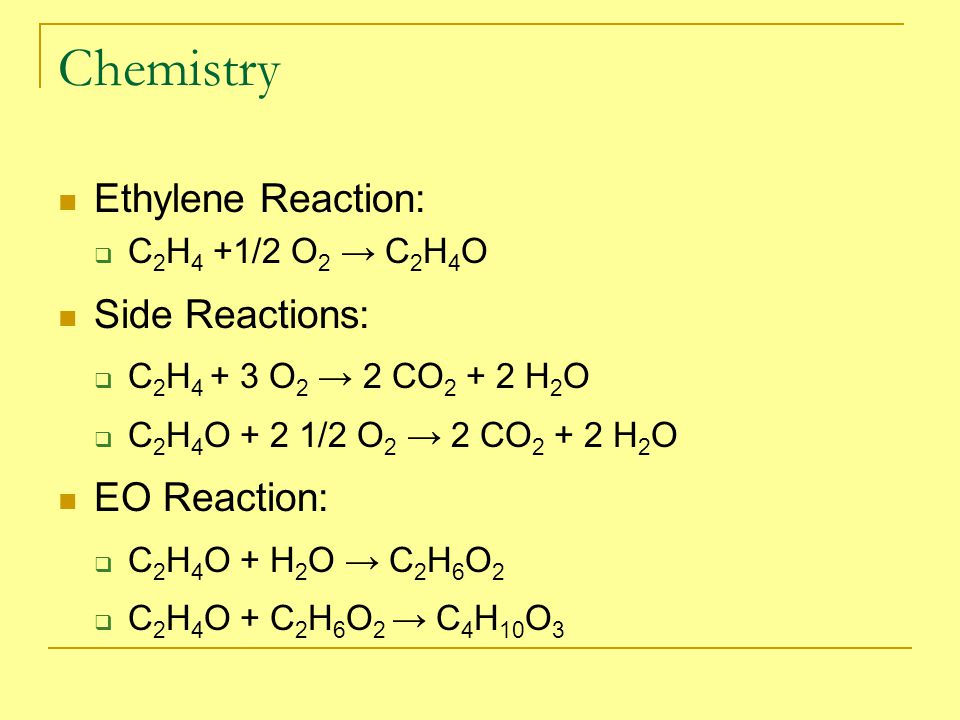
SOLVED: Section 2: Chemical reactions and stoichiometry Q2. Ethylene glycol (C2H6O2) can be produced from the reaction of ethylene oxide (C2H4O) and water as shown below: C2H4O(g) + H2O(g) -> C2H6O2(g) If

Numerical thermal study on CNTs/ C2H6O2– H2O hybrid base nanofluid upon a porous stretching cylinder under impact of magnetic source - ScienceDirect

Ethylène glycol, diol, molécule C2H6O2. Il : image vectorielle de stock (libre de droits) 1718457154 | Shutterstock

PDF) Thermal Transport Investigation in Magneto- Radiative GO-MoS2/H2O- C2H6O2 Hybrid Nanofluid Subject to Cattaneo-Christov Model