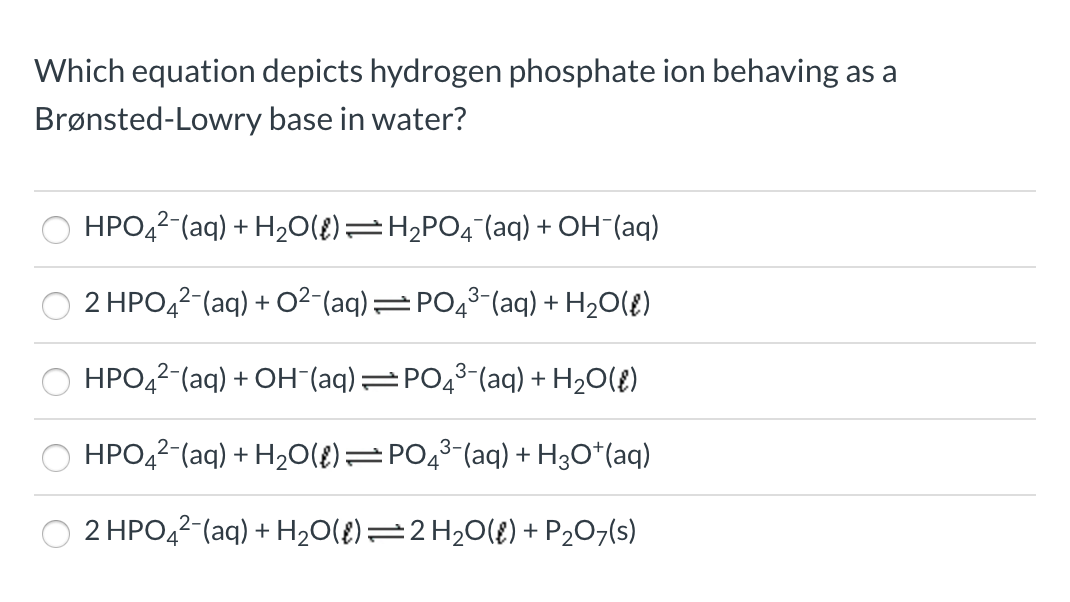![Na2[(VO)2(HPO4)2(C2O4)]·2H2O: A Promising Mixed Polyanionic Cathode Material for Aqueous Zn-Ion Batteries Na2[(VO)2(HPO4)2(C2O4)]·2H2O: A Promising Mixed Polyanionic Cathode Material for Aqueous Zn-Ion Batteries](https://pubs.acs.org/cms/10.1021/acs.inorgchem.2c03308/asset/images/medium/ic2c03308_0004.gif)
Na2[(VO)2(HPO4)2(C2O4)]·2H2O: A Promising Mixed Polyanionic Cathode Material for Aqueous Zn-Ion Batteries

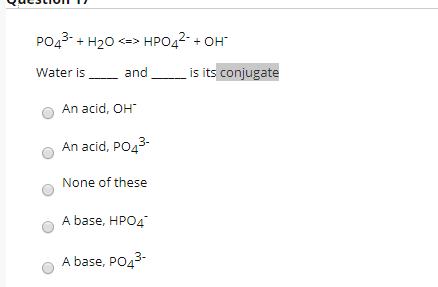
SOLVED: Choose balanced equation for the transfer of a proton between dihydrogen phosphate ion and the hydroxide ion: H2PO4-(aq) + OH-(aq) â†' HPO4 2-(aq) + H2O(l)

Catalytic conversions of isocyanate to urea and glucose to levulinate esters over mesoporous α-Ti(HPO4)2·H2O in green media - X-MOL

Figure 6 from VIIIVIV(HPO4)4·enH·H2O: a mixed-valence vanadium phosphate with an open framework | Semantic Scholar
![Ca2[Ti(HPO4)2(PO4)]·H2O, Ca[Ti2(H2O)(HPO3)4]·H2O, and Ti(H2PO2)3: Solid-State Oxidation via Proton-Coupled Electron Transfer | Inorganic Chemistry Ca2[Ti(HPO4)2(PO4)]·H2O, Ca[Ti2(H2O)(HPO3)4]·H2O, and Ti(H2PO2)3: Solid-State Oxidation via Proton-Coupled Electron Transfer | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/acs.inorgchem.1c02685/asset/images/large/ic1c02685_0008.jpeg)
Ca2[Ti(HPO4)2(PO4)]·H2O, Ca[Ti2(H2O)(HPO3)4]·H2O, and Ti(H2PO2)3: Solid-State Oxidation via Proton-Coupled Electron Transfer | Inorganic Chemistry
Synthesis and structural characterisation of solid titanium(IV) phosphate materials by means of X-ray absorption and NMR spectro

Single-crystal neutron diffraction and Mössbauer spectroscopic study of hureaulite, (Mn,Fe)5(PO4)2 (HPO4)2 (H2O)4 - European Journal of Mineralogy Volume 28 Number 1 — Schweizerbart science publishers

SOLVED: The following reaction is taking place in aqueous solution: HPO42- + H2O -> H3O+ + PO43-. Which is a Bronsted-Lowry base in this reaction? Select one: a. H+ b. H2PO4- c.

Figure 5. XRD of Zr0.8Ti0.2(HPO4)2.H2O : α- Zirconium Titanium Phosphates - Fibrous Cerium Phosphate Composite Membranes and Their 1,10- Phenanthroline Cu(II) Pillared Materials : Science and Education Publishing


![Answered: What is the [HPO4-2] of a solution… | bartleby Answered: What is the [HPO4-2] of a solution… | bartleby](https://content.bartleby.com/qna-images/answer/25d6589c-34c4-49fd-a0de-f621c4bd4dbc/9f74c13f-f553-447b-8735-1f1f7effb02f/x03vih.png)