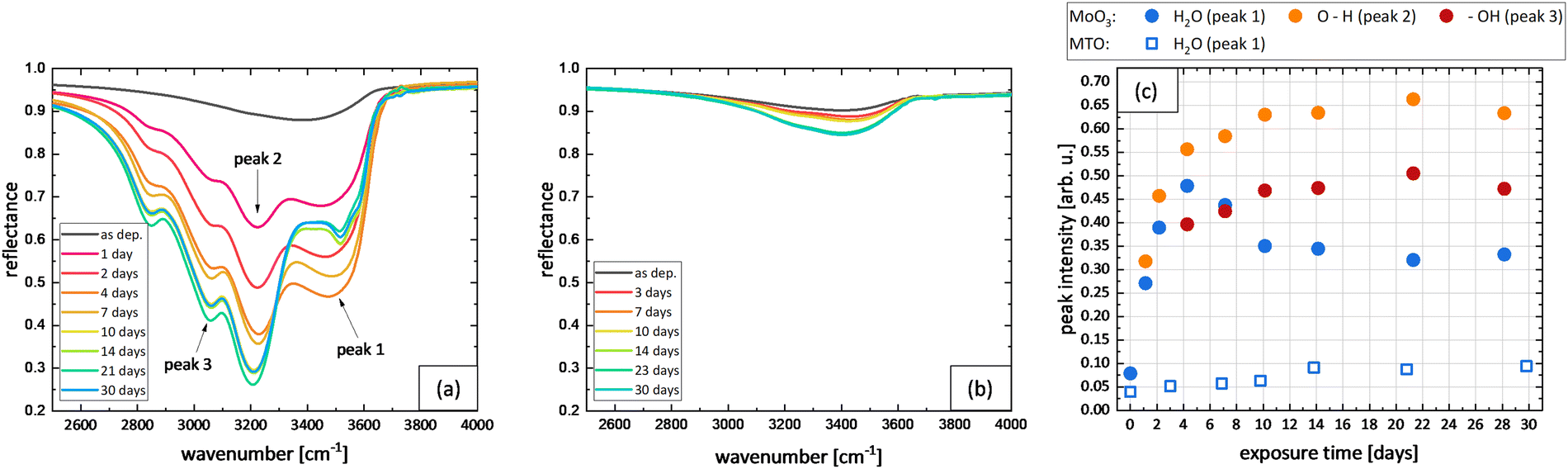
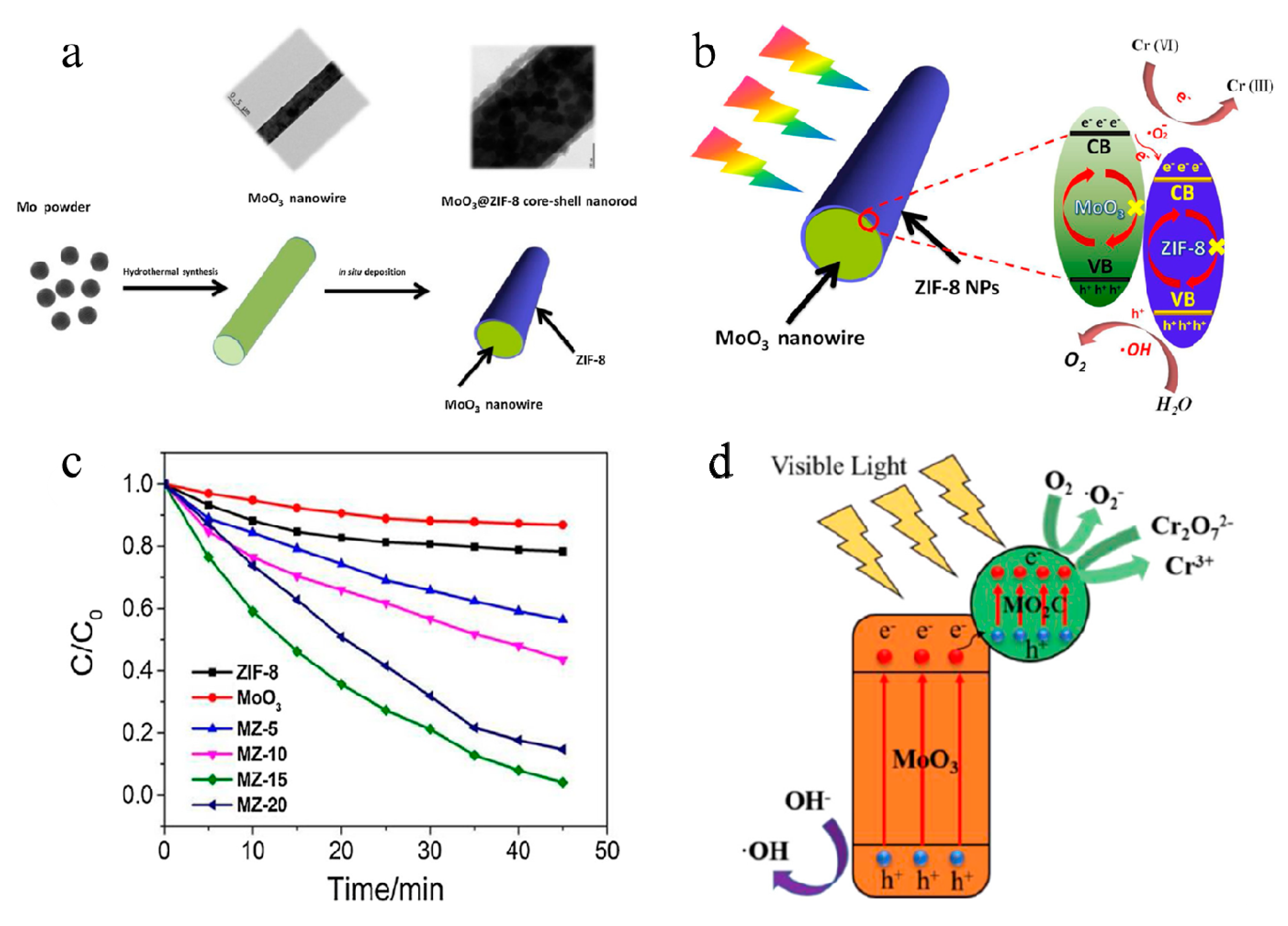
Aqueous synthesis of molybdenum trioxide (h-MoO3, α-MoO3·H2O and h-/α-MoO3 composites) and their photochromic properties study - ScienceDirect
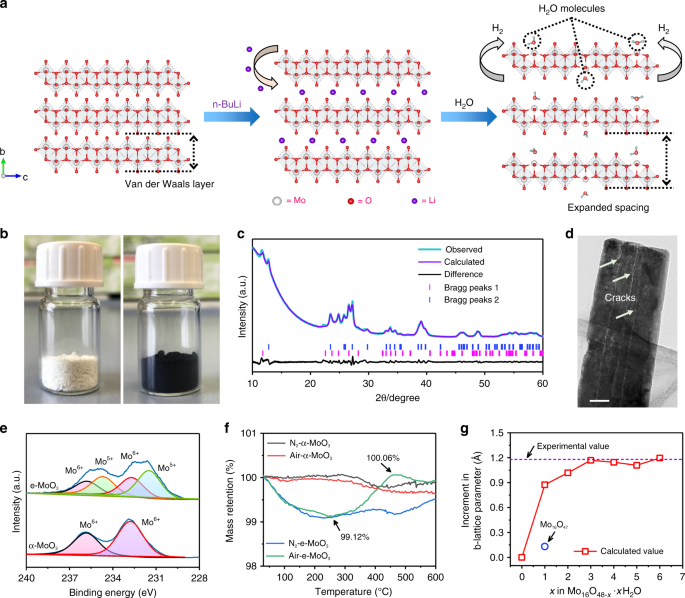
Interlayer gap widened α-phase molybdenum trioxide as high-rate anodes for dual-ion-intercalation energy storage devices | Nature Communications

Single Crystals of α-MoO3-Intercalated {Ni(H2O)6}2+ and Electrocatalytic Water Reduction: Toward a Class of Molybdenum Bronzes | Inorganic Chemistry
![PDF] Photoluminescence and Raman Studies of α-MoO3 Doped with Erbium and Neodymium | Semantic Scholar PDF] Photoluminescence and Raman Studies of α-MoO3 Doped with Erbium and Neodymium | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/1183ded5d8e0e68d0ac92fa6c3c408f9554eb1bb/5-Figure5-1.png)
PDF] Photoluminescence and Raman Studies of α-MoO3 Doped with Erbium and Neodymium | Semantic Scholar

Charge redistribution for MoO3-H2O (a), MoO3-4H2O (b), and VO-MoO3-H2O | Download Scientific Diagram
PDOS of perfect-MoO3 (a), MoO3-H2O (b), MoO3-4H2O (c), VO-MoO3 (d), and | Download Scientific Diagram
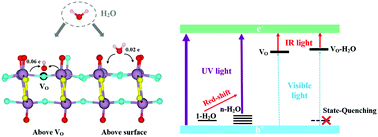
Oxygen deficient α-MoO3 with enhanced adsorption and state-quenching of H2O for gas sensing: a DFT study - Journal of Materials Chemistry C (RSC Publishing)

Nanoporous MoO3−x/BiVO4 photoanodes promoting charge separation for efficient photoelectrochemical water splitting | Nano Research

Room-temperature synthesis and solar photocatalytic performance of MoO3·0.5 H2O nanorods - ScienceDirect

Hydrophobic/Hydrophilic Interplay in 1,2,4‐Triazole‐ or Carboxylate‐Based Molybdenum(VI) Oxide Hybrids: A Step Toward Development of Reaction‐Induced Self‐Separating Catalysts - Lysenko - 2021 - ChemCatChem - Wiley Online Library