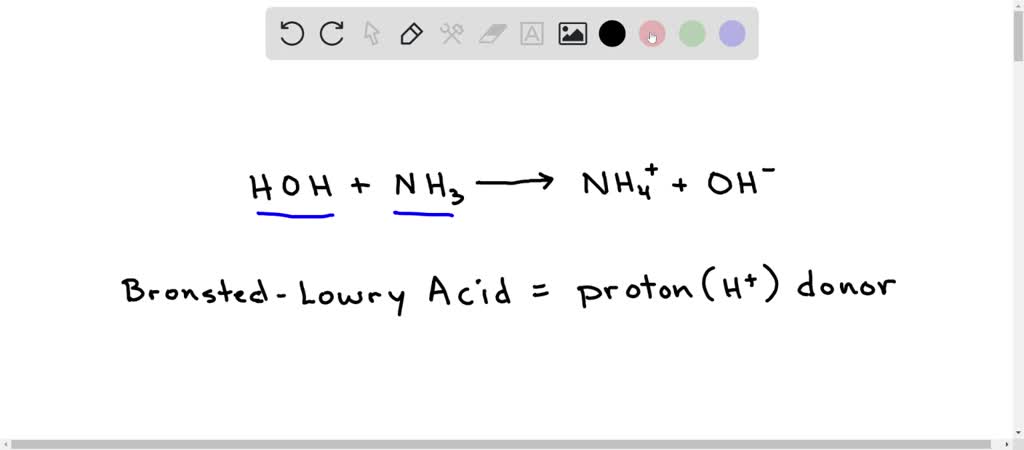
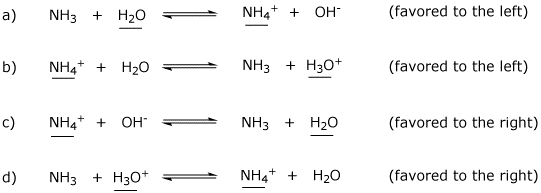
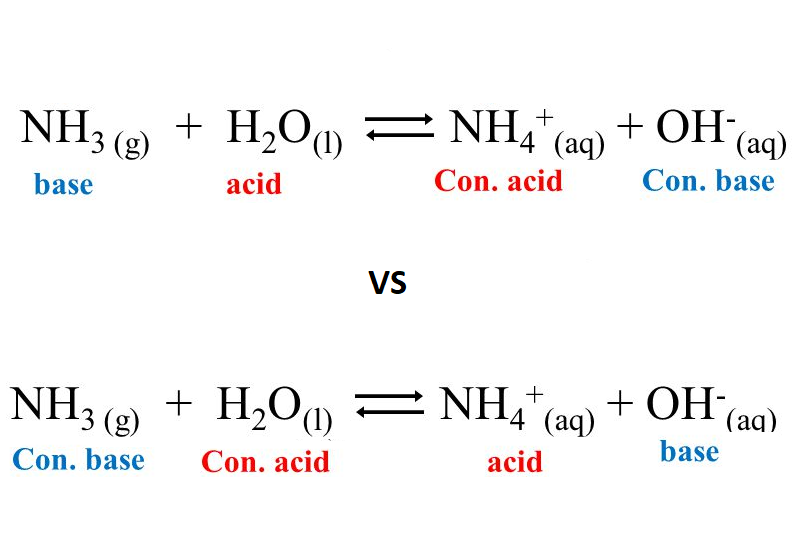
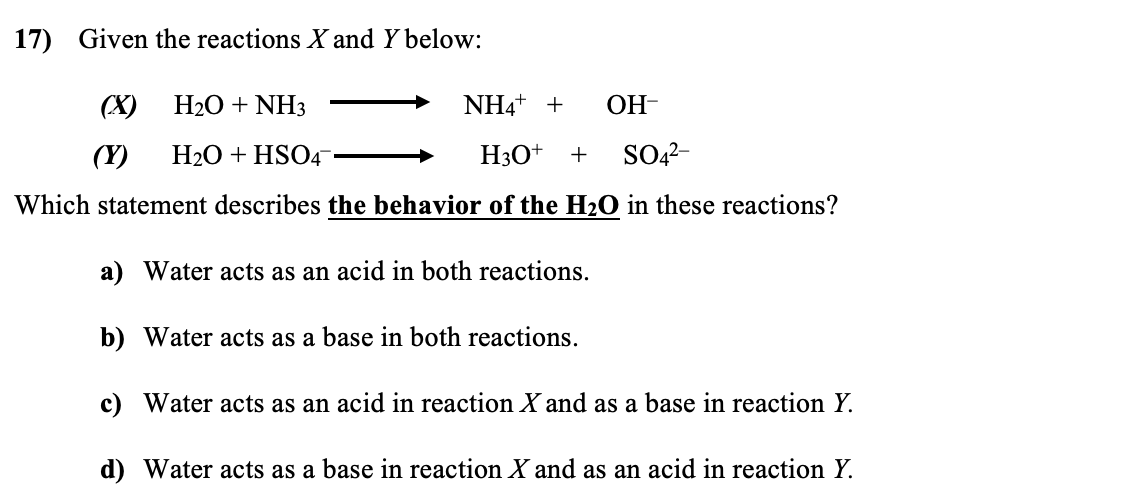
OneClass: Consider this reaction: NH4OH(aq) rightarrow NH3(aq) + H2O(aq) At a certain temperature it ...
Memoire Online - Optimisation d'une machine frigorifique à absorption-diffusion nh3-h2o-h2 - Souad Himoun
Catalytic effect of (H2O)n (n = 1–3) on the HO2 + NH2 → NH3 + 3O2 reaction under tropospheric conditions - RSC Advances (RSC Publishing)

L'approvisionnement (AMH) de NH3· H2O Prix d'hydroxyde d'ammonium - Chine Hydroxyde d'ammonium, l'Ammoniac L'EAU

L'approvisionnement (AMH) de NH3· H2O Prix d'hydroxyde d'ammonium - Chine Hydroxyde d'ammonium, l'Ammoniac L'EAU

Modèle Chimique Des Molécules D'ammoniac Nh3 Eau H2o Méthane Ch4 Illustration Vectorielle Isola | Vecteur Premium

Several isomers are possible for (Co(en)(NH3)(H2O)2Cl)2+. How many isomers can you draw for this complex? | Homework.Study.com

Effect of water and ammonia on the HO + NH3 → NH2 + H2O reaction in troposphere: Competition between single and double hydrogen atom transfer pathways - ScienceDirect

NH3⋅H2O: The Simplest Nitrogen‐Containing Ligand for Selective Aerobic Alcohol Oxidation to Aldehydes or Nitriles in Neat Water - Zhang - 2018 - ChemistryOpen - Wiley Online Library
![The CO2-NH3-H2O system as described by the Thomsen model [7]. | Download Scientific Diagram The CO2-NH3-H2O system as described by the Thomsen model [7]. | Download Scientific Diagram](https://www.researchgate.net/publication/319196113/figure/fig1/AS:533643246669824@1504241872081/The-CO2-NH3-H2O-system-as-described-by-the-Thomsen-model-7.png)